Chief Electrocoat
Contact UsHere’s What you Can Expect
WEATHER RESISTANCE
The Chief Electrocoat primer is superior to spray on primers most commonly found within our industry. It penetrates where others can’t because the steel is fully submerged in an aqueous solution during application.
DURABLE FINISH
Because the coating is electro-statically applied and then oven baked, the resulting enamel finish is durable and will withstand loading, shipping and jobsite handling.
GOOD IDEA
When compared to pre-coated sheet steel, our electrocoat is superior in the fact that all welding and necessary holes are completed prior to the primer application.
ECONOMICAL ADVANTAGES
For most interior applications, the primed steel can be left as is for many years and still exhibit a glossy finished appearance. Most top coats can be applied directly over the primer, saving you time and money.
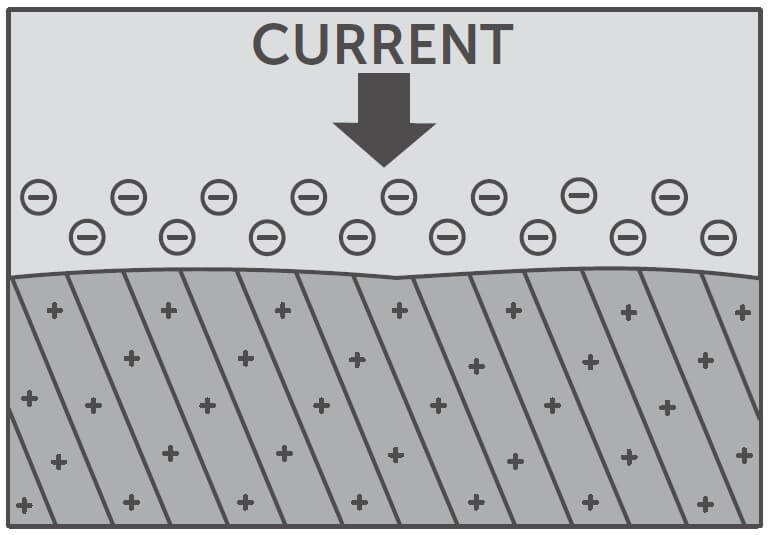
HOW ELECTROCOATING WORKS
Electrocoating is a process in which we apply gray oxide primer to our structural steel members to prevent corrosion. This process, superior to any spray-on application, allows us to completely coat the steel with primer, even in the tightest corners. There is no other process in the industry that gives the same consistent result.
At its simplest, here’s what you need to know: Electrocoating is aptly named, as the process involves using electrical currents to create a protective coat on the steel.
The result is a visually appealing, smooth gray finish that is water resistant. Better yet, it’s paintable. Add a finish coat and enjoy the look you deserve.